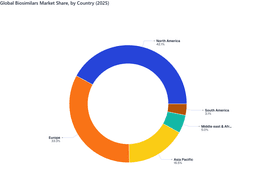
The Biosimilars Market is growing rapidly due to patent expirations of major biologics, affordable therapeutic alternatives, and increasing use in oncology, autoimmune disorders, and chronic diseases. The global market stands at USD 36,685.84 million in 2025 and is projected to reach USD 84,069.34 million by 2032, growing at a CAGR of 12.35%. Rising demand for monoclonal antibodies, erythropoietin, and insulin biosimilars supports this growth. North America leads the Biosimilars market in 2025, contributing USD 15,431.01 million, while Asia-Pacific and South America are high-growth regions. Pricing, regulatory requirements, and operational factors shape the Biosimilars Market. Agencies such as the FDA, EMA, and WHO enforce rigorous comparability studies and post-marketing surveillance, which increases development timelines but strengthens clinician confidence. Efficient manufacturing, optimized cell-line engineering, and improved antibody production reduce costs. Challenges include interchangeability hurdles, complex supply chains, and high development costs, but growing acceptance of therapeutic equivalence continues to drive adoption.
Biosimilars overview
Published by MMR Statistics Research Team, Updated